GENE TRANSFER METHODS
The challenge in gene therapy is to deliver an adequate amount of genetic material into target cells or tissues and to maintain gene expression for a desired period of time. Genetic material can be introduced to their target cells or tissues via different methods of delivery. In principle, we can group them into: (1) physical; (2) viral; (3) non-viral methods; and (4) bacterial or yeast.
Electroporation, ultrasound, and gene gun deliveries are examples of physical methods that have been used. As the name already implies, with viral vectors a biological (i.e., virus) vector is used as a vehicle to deliver the genetic material into the cells, whereas with non-viral gene transfer methods a synthetic carrier (liposomes or nanoparticles) is used. Different vectors have different properties in relation to their transduction efficiency and their efficacy to express the introduced genes. In addition, they differ in respect of the duration of expression of the transgene, as well as their safety profile.
Depending on the requirements, different vectors can be used for different therapeutic purposes. Currently, viral vectors are considered as the most effective of all gene delivery methods for in vivo gene transfer. Ideally, the gene transfer vector should be able to target a specific tissue with high transduction efficiency and sustain a stable, regulated gene expression without any side effects or immunogenic responses. Unfortunately, none of the currently used gene delivery vectors fulfil all these criteria. Local injection of a vector typically results in a limited, but accurate effect area.[7]
Electroporation, ultrasound, and gene gun deliveries are examples of physical methods that have been used. As the name already implies, with viral vectors a biological (i.e., virus) vector is used as a vehicle to deliver the genetic material into the cells, whereas with non-viral gene transfer methods a synthetic carrier (liposomes or nanoparticles) is used. Different vectors have different properties in relation to their transduction efficiency and their efficacy to express the introduced genes. In addition, they differ in respect of the duration of expression of the transgene, as well as their safety profile.
Depending on the requirements, different vectors can be used for different therapeutic purposes. Currently, viral vectors are considered as the most effective of all gene delivery methods for in vivo gene transfer. Ideally, the gene transfer vector should be able to target a specific tissue with high transduction efficiency and sustain a stable, regulated gene expression without any side effects or immunogenic responses. Unfortunately, none of the currently used gene delivery vectors fulfil all these criteria. Local injection of a vector typically results in a limited, but accurate effect area.[7]
GENE THERAPY VECTOR SYSTEMS
An important feature for any successful gene therapy protocol is the vector system. To date, a number of vectors have been developed, including both viral and non-viral based therapy systems .These vectors possesses a number of unique features and each has its own advantages and disadvantages. The most promising existing vectors are the replication-competent oncolytic viral vector based gene therapy systems, particularly the adenoviruses.[8]
VIRAL VECTOR SYSTEMS
|
Adenoviral vectors Adenovirus vector (AV) is the most commonly studied and most widely used system in cancer gene therapy. It is of particular use for cancer gene therapy applications, where temporary gene expression is acceptable or even beneficial. There are several serotypes, but the currently employed AVs in clinical trials are mostly based on serotype 5. These vectors can replicate highly and have demonstrated efficient gene transfer into various types of cancer cells. Several other adenoviruses, based on canine, porcine, bovine, ovine and avian adenoviruses have been developed. Unfortunately, AVs contain many viral genes which encode for major proteins that elicit a strong host immune response. Of particular concern is the release of cytotoxic T lymphocytes that lyse cells expressing the recombinant genes. Newer generations of AV vector have been designed to overcome some of these problems and initial results are encouraging.
|
New techniques involved in removing the recombinant viral genes and transfecting the non-recombinant plasmid with a helper virus and then separating the helper virus with sedimentation techniques have been developed. Improvements in helper virus have also been trialled that reduces "floxed" helper virus production 1000-fold, but this method still has a 1% wide type (WT) contamination thus still allowing the possibility of in vivo recombination. With regard to AV-mediated cancer treatment, high-level tumour transduction remains a key developmental hurdle. To this end, AV vectors possessing infectivity enhancement and targeting capabilities should be evaluated in the most stringent model systems possible. Advanced AV-based vectors with imaging, targeting and therapeutic capabilities have yet to be fully realized; however, the feasibilities leading to this accomplishment are within close reach.[8]
|
Adeno-associated virus (AAV)-based vectors AAV-based vectors have been shown to be non-toxic and undergo widespread cellular uptake in preclinical evaluation. A study has compared five different AAV strains and amongst them, serotype 2 was proven to be the most efficient killers of tumour cells. In another study, serotype 8 AAV vector encoding a soluble vascular endothelial growth factor (VEGF) receptor was able to halt tumour growth in several rodent glioma models. However, difficulties in the development of packaging cell lines for AAV, as well as bulk production and vector purification have been reported as problematic. A new system was developed recently to scale up and bulk production of AAV from insect cells which may solve some of these existing problems.[8]
|
Herpesvirus-based vectors Vectors based on herpesviruses are well-developed and have progressed to clinical trials. As with other viral vectors, replication-defective vectors have not shown much potential. The first replication competent vector was based on a mutant strain, where the vectors are deleted from the main neurovirulence gene r34.5, thus restricting its ability to replicate in adult central nervous system and to form latency. However, later study showed that the mutant strain which had the deletion of the r34.5 gene also had reduced capacity for replication inside tumour cells. Pre-existing immunity may pose a problem that limits the clinical efficacy of herpesvirus based vectors. This immunity prevents the transduction of peripheral organs and also can cause liver toxicity. However, a recent mutant strain-secreting cytokine granule macrophage colony stimulatory factor (GM-CSF) or IL-12 was shown to be effective in liver cancer therapy in a murine model which likely involves both direct viral oncolysis and actions of specific immune effector cells.[8]
Viral replicons and transposons Semliki Forest virus (SFV) subgenomic replicons (i.e. non toxic replication) have been developed that allow stable expression of a required gene e.g. beta-galactosidase (beta-Gal) in mammalian cell lines. Studies showed that expression remained high (approximately 150 pg per cell) throughout cell passages. Potential problems with the use of transposons for gene therapy may arise from having no 'off' switch for the transposase and the relatively low quantities of integrated product, either of which would make retroviral intergrase a more suitable or alternative enzyme for chromosomal integration.[8]
Targeted viral vectors While efforts have been focused on the continuing refinement of various vector systems, several obstacles remain, primarily the low efficiency of gene delivery into target tumour cells. The vascular endothelial wall is a significant physical barrier prohibiting access of systemically administered vectors to the tumour cell. To overcome this obstacle, strategies are currently being developed to take advantage of transcytosis pathways through the endothelium. An AV vector targeted to the transcytosing transferrin receptor pathway, using the bifunctional adapter molecule has been constructed. The transcytosed AV virions retain the ability to infect cells, establishing the feasibility of this approach. However, efficiency of AV trafficking via this pathway is poor. Other efforts are directed towards exploring other transcytosing pathways such as the melanotransferrin pathway, the poly-IgA receptor pathway, or caveolae-mediated transcytosis pathways.[8]
Targeted viral vectors While efforts have been focused on the continuing refinement of various vector systems, several obstacles remain, primarily the low efficiency of gene delivery into target tumour cells. The vascular endothelial wall is a significant physical barrier prohibiting access of systemically administered vectors to the tumour cell. To overcome this obstacle, strategies are currently being developed to take advantage of transcytosis pathways through the endothelium. An AV vector targeted to the transcytosing transferrin receptor pathway, using the bifunctional adapter molecule has been constructed. The transcytosed AV virions retain the ability to infect cells, establishing the feasibility of this approach. However, efficiency of AV trafficking via this pathway is poor. Other efforts are directed towards exploring other transcytosing pathways such as the melanotransferrin pathway, the poly-IgA receptor pathway, or caveolae-mediated transcytosis pathways.[8]
Viral vector-associated multifunctional particles (MFPs) Nanotechnology has recently been incorporated into viral vector systems in the form of multifunctional particles (MFPs). Nanotechnology is defined as the development of devices of 100 nm or smaller, having unique properties due to their scale. The devices that are
developed generally incorporate inorganic or biological material. In thisregard the coupling of inorganic nano-scale materials to targeted AV vectors has much potential for tumour targeting, imaging and amplified tumour killing capacities. For example, magnetic nano-particles have recently received much attention due to their potential application in clinical cancer treatment; targeted drug delivery and magnetic resonance imaging (MRI) contrast agents.[8]
developed generally incorporate inorganic or biological material. In thisregard the coupling of inorganic nano-scale materials to targeted AV vectors has much potential for tumour targeting, imaging and amplified tumour killing capacities. For example, magnetic nano-particles have recently received much attention due to their potential application in clinical cancer treatment; targeted drug delivery and magnetic resonance imaging (MRI) contrast agents.[8]
|
Oncolytic virus An oncolytic virus is a virus that has the ability to infect cancer cells and cause oncolysis. It is with obvious reasons that these types of viruses have received much attention in the field of cancer therapy as they can result in direct destruction of the tumour cells. Recent advances in genetic modification techniques of oncolytic viruses have again awoken researchers’ interest into these agents for cancer therapy. There are now many different oncolytic viruses currently being trailed as potential therapeutic agents. The first to be produced for the clinic was ONYX-015 which has been shown to be a safe anticancer agent.[8]
|
LIMITATIONS OF VIRAL VECTORS
Although both viral vectors and oncolytic viruses are widely used in cancer gene therapy research, these vectors suffer from several limitations.
The first of these is the virus’s inability to specifically seek out and target the tumour. The majority of therapies that utilize these vectors currently require intratumoural injection to elicit an effect. Whilst this approach may be beneficial in some cases, its uses are limited as many tumours are inaccessible and may have already spread to other areas of the body at the time of diagnosis and treatment, making them difficult to locate and treat.
Another deficiency of viral vectors is their lack of capacity to efficiently penetrate and kill every cell within the tumour mass. This results in non-tumour stromal cells being unaffected thus having the ability to regrow into another tumour. Finally, the tumours hypoxic microenvironment also reduces the effectiveness of the viral vector.[8]
The first of these is the virus’s inability to specifically seek out and target the tumour. The majority of therapies that utilize these vectors currently require intratumoural injection to elicit an effect. Whilst this approach may be beneficial in some cases, its uses are limited as many tumours are inaccessible and may have already spread to other areas of the body at the time of diagnosis and treatment, making them difficult to locate and treat.
Another deficiency of viral vectors is their lack of capacity to efficiently penetrate and kill every cell within the tumour mass. This results in non-tumour stromal cells being unaffected thus having the ability to regrow into another tumour. Finally, the tumours hypoxic microenvironment also reduces the effectiveness of the viral vector.[8]
NON-VIRAL VECTOR SYSTEMS
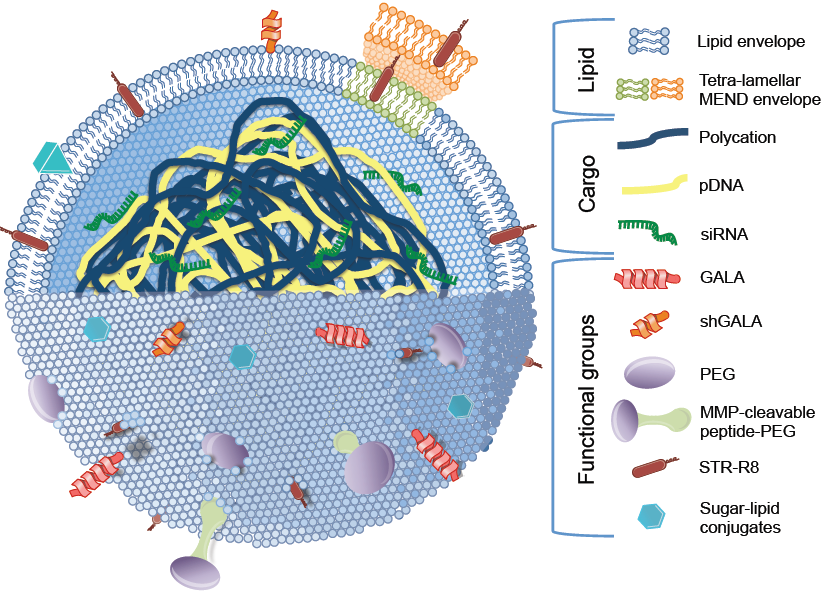
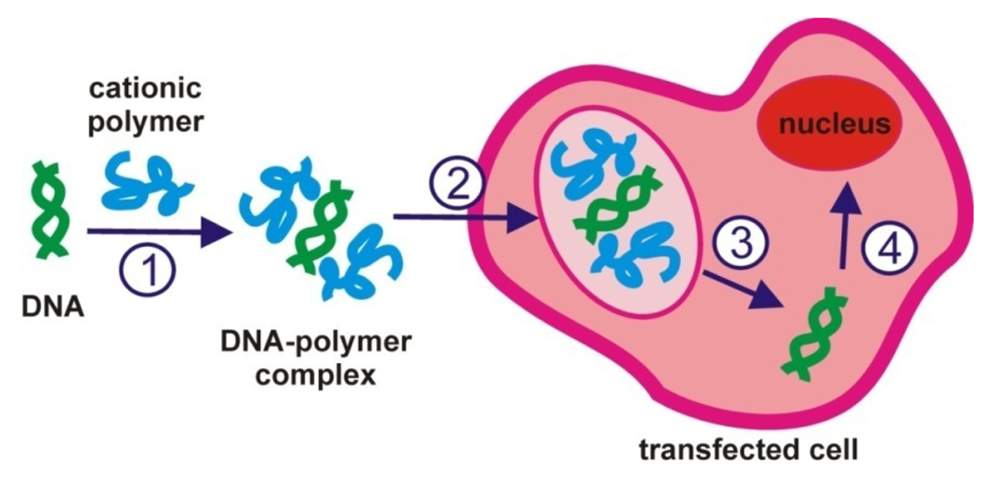
The multiple advantages of non-viral vectors has attracted special focus to derive more sophisticated molecules that can be used for the appropriate delivery of the nucleic acids without imposing the threats of the conventional viral vectors. These non-viral vectors owe its therapeutic advantage to the properties possessed such as low toxicity and comparatively low immunogenicity. Apart from this the presence of the physicochemical versatility that enables these molecules to modify their surface for targeting specific cells/tissues.[9]
|
Lipid-based nanoparticles Design of these carrier molecules ranges from dendrimers, nanogold, liposomes, nanoemulsions, drug-carrier conjugates etc. are based upon the assemblies of naturally/synthetically occurring components. Lipid based nanoparticles are very promising in the delivery of nucleic acids at the desired region. One of the best activity can be observed with liposome-mediated intratumoral delivery of genes that code for interferon proteins, IFN-β which in turn generates an anti tumoral activity. Similarly cationic liposomes carrying the IFB-β gene into the tumour region of the patients has resulted in a considerable decrease in the tumour volume. Moreover the incorporation of the gene was well endured. This study was conducted on a very small scale thus no significant conclusion was drawn from it. However such therapy can be studied to determine the immunogene response on a large scale that can be effectively used for GBM.[9]
|
|
Polymer based carrier systems Polymer based nanoparticles finds application in anticancer therapies because of their multifunctionality and specificity. These molecules can carry nucleoside analogues that are very effective in anticancer procedures. Nucleoside analogues because of the antimetabolite nature and their ability to interfere with the synthesis of nucleic acids in cellular and cancer cell systems, there is an immediate need to design molecules that can effectively carry these analogues to the desired location. Usually a high dose of these nucleoside analogues is required because of their nonspecific distribution as well as rapid clearance from the system.[9]
|
Neural & mesenchymal stem cells as carrier molecules Stem cells have tremendous application as carrier molecules because of their highly specific nature to target cancer cells (gliomas).The underlying concept is the secretion of various cytokines and growth factors such as Vascular Endothelial (VEGF) and Platelet- Derived Growth Factors (PDGF). These molecules acts as chemo attractants that drives the migration of these cells towards the target tumour. Neural and mesenchymal stem cells in particular, are being generally employed for the aforementioned procedure. Adipose tissue and bone marrow are very rich source of Mesenchymal Stem Cells (MCS’s) whereas both adult and foetal brain is good source of neural stem cells.[9]
Mesenchymal stem cells have certain advantages that enable them to be used as a most efficient therapeutic delivery agent. The tumortropic property of these cells makes them accumulated at the cancer site thereby enabling better delivery of the therapeutic agent. The unique ability to modulate the immune response increases the possibility of survival of MCS’s. Moreover the potential application of using autologous stem cells minimizes the chances of immune response, if any, thereby increasing the cell survival.[9]
Mesenchymal stem cells have certain advantages that enable them to be used as a most efficient therapeutic delivery agent. The tumortropic property of these cells makes them accumulated at the cancer site thereby enabling better delivery of the therapeutic agent. The unique ability to modulate the immune response increases the possibility of survival of MCS’s. Moreover the potential application of using autologous stem cells minimizes the chances of immune response, if any, thereby increasing the cell survival.[9]
DECLAIMER: This site is made for a academic purpose, do not consider it for a professional use.